Achieved positive adjusted EBITDA in Q4 2025 and near break-even $(0.29) million for the full year
Advancements across business units, R&D, and clinical development to support scale and international growth
TORONTO, April 01, 2026 (GLOBE NEWSWIRE) -- Avicanna Inc. (“Avicanna” or the “Company”) (TSX: AVCN) (OTCQX: AVCNF) (FSE: 0NN) a biopharmaceutical company focused on the development, manufacturing and commercialisation of plant-derived cannabinoid-based products is pleased to announce that its full year 2025 results, audited financial statements with management’s discussion and analysis have been filed.
“Throughout the year, we strengthened our leadership in cannabinoid-based medicine by advancing our research and development programs, progressing clinical initiatives, and deepening engagement with the Canadian medical community. We delivered the strongest financial performance in our history, reflecting operational discipline, optimization, and efficiencies.
“We also made targeted investments across our platforms to support commercial scalability, advanced product development, and enhanced the capabilities of the MyMedi.ca platform. These initiatives position Avicanna to scale efficiently, expand internationally, and unlock the full value of our business units and intellectual property globally,” stated Aras Azadian, CEO of Avicanna.
Earnings call: Management will host an earnings call and corporate update on Tuesday April 7th at 8:00 a.m. Eastern time. Presenters will include Aras Azadian, CEO, Nick Hillborn, Interim CFO and Dr. Karolina Urban, EVP, Medical Affairs. To register, follow the link: https://zoom.us/webinar/register/WN_RJACfyrPSo6CpVfSzcx5SQ
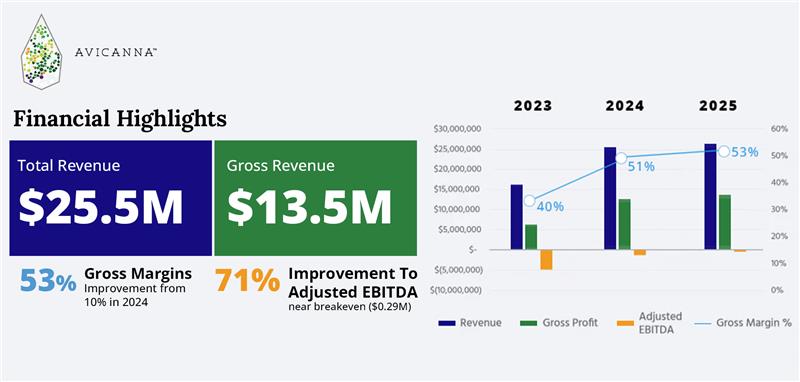
Financial Highlights:
- Revenue and Gross Profit: Revenue totaled $6.6 million in Q4 2025 and $25.48 million for the year ended December 31, 2025, consistent with $6.6 million and $25.46 million, respectively, in the prior year. Gross margin expanded to 53% in 2025 from 51% in 2024, driving gross profit to $13.4 million, compared to $12.9 million in the prior year. Margin expansion reflects continued optimization of product mix and cost efficiencies.
- Adjusted EBITDA: The Company generated positive adjusted EBITDA of $0.31 million in Q4 2025, compared to $(0.79) million in Q3 2025, reflecting improved operating leverage. For the year ended December 31, 2025, the Company achieved near break-even adjusted EBITDA with $(0.29) million, representing a 76% improvement compared to $(1.01) million in 2024. The year-over-year improvement was primarily attributable to higher gross margins and a 10% reduction in operating expenses.
Highlights and Advancements During 2025:
Canadian Commercial Advancements: During 2025 the Company further advanced its Canadian commercial platform across new SKUs, channels and listings. Additionally, the Company’s MyMedi.ca platform recovered from a modest Q1 performance of decreased sales, with three consecutive quarters of growth, combined with consistent increases in the proportion of sales attributed to Avicanna’s proprietary products. This resulted in 211,090 units of proprietary products sold during 2025, an increase of approximately 5% from 2024. At the end of the fourth quarter, the Company had 52 commercial SKUs and 174 commercial listings across medical and adult use channels, representing 27% growth in proprietary commercial SKUs and a 32% growth in total listings from Q4 2024.
Initiation of Pilot Phase II Clinical Study for Osteoarthritis Pain with UHN: The multicenter, blinded, randomized placebo-controlled investigator-initiated study, will be led by Dr. Hance Clarke and conducted at University Health Network. The study will analyze the effectiveness of Avicanna’s proprietary oral cannabis extracts for osteoarthritic pain and is the Company’s first placebo controlled, blinded randomized multicenter trial “RCT”. The study is funded by a Canadian Institutes of Health Research (“CHIR”) grant and is sponsored by Avicanna, which is providing its proprietary CBD and THC capsules produced under Good Manufacturing Practices (“GMP”) for the trial.
Promising Pre-clinical Data Supporting Enhanced Absorption and Patent Filing for Novel Oral Delivery Platform: The PwdRx technology was designed to address key formulation challenges associated with highly lipophilic cannabinoids, which often exhibit poor water solubility, low and variable bioavailability, and delayed onset of action. The technology demonstrated positive results in recent in-vitro study including 74% higher bioavailability (AUC), 63% faster peak plasma levels (TMAX) and a 134% higher peak plasma concentration (CMAX) when compared to an MCT oil formulation. The Company also filed a provisional patent application on the novel drug delivery platform which can be incorporated into products such as tablets, capsules, sachets and pouches.
Scientific and Medical Affairs Collaboration with Aspeya Switzerland SA: The scientific and medical affairs collaboration with Aspeya Switzerland SA (formerly Vectura Fertin Pharma) a subsidiary of Philip Morris International, aims to facilitate research and medical affairs initiatives related to medical cannabis in Canada. The scientific and medical affairs collaboration will prioritize engagement with the Canadian medical community, patients, patient advocacy groups, and insurers to gain insights into the challenges associated with accessing medical cannabis. The scientific and medical affairs collaboration will leverage Avicanna’s medical cannabis platform, including MyMedi.ca, with the aim of improving patient access and patient support.
Symposium on Cannabinoid-based Medicine (June 2025): Held at the MaRS Discovery District in Toronto, Avicanna’s fifth symposium brought together leading key opinion leaders and healthcare professionals to explore advancements in cannabinoid-based research, medicine, and clinical adoption. The event’s attendees included healthcare practitioners and researchers, and featured discussions on emerging evidence and practical clinical applications of cannabinoid-based medicine. The program showcased insights from prominent clinicians, researchers, scientists, and industry experts representing a range of academic, clinical, and research institutions.
First Commercial Exports of Premium and Organic Cannabis Flower: Santa Marta Goldan Hemp SAS (“SMGH”), the Company’s majority owned subsidiary, completed its first export of organic certified flower into Switzerland during Q3 2025 and its first commercial export of organic flower to Australia in Q1 2026. The accumulated exports represent the 22nd international market for SMGH and 24 market for Avicanna. These new commercial opportunities are the result of improvements to the SMGH infrastructure and expansion of the Aureus portfolio to meet the growing demand of medical cannabis flower in Europe and Australia.
Avicanna LATAM SAS Launches re+PLAY™ CBD Wellness Brand Topicals with Harrington Wellness Brands in the United States: re+PLAY™ is a CBD wellness brand founded by NBA veteran Al Harrington that features Avicanna’s patented and proprietary CBD and CBG topical formulations. Initial product offerings include a 3% CBD localized cream and the 2% CBD and 1% CBG transdermal gel employing Avicanna’s patented deep tissue technology. The CBD and CBG used in the formulations are derived from USDA organic certified hemp cultivated in Avicanna’s subsidiary SMGH and manufactured by the Avicanna LATAM SAS team in Colombia.
About Avicanna:
Avicanna is an international biopharmaceutical company specializing in the commercialization of proprietary and evidence-based cannabinoid-based products for the global medical and pharmaceutical market segments. Avicanna has established scientific and medical affairs platforms that support its four commercial stage business pillars and have resulted in the commercialization of more than fifty finished products.
- Medical Cannabis formulary (RHO Phyto™): The formulary offers a diverse range of proprietary products including oral, sublingual, topical, and transdermal deliveries with varying ratios of cannabinoids, supported by ongoing patient and medical community education. RHO Phyto™ is an established brand in Canada currently available nationwide across several channels and expanding into new international markets.
- Medical cannabis care platform (MyMedi.ca): MyMedi.ca is a medical cannabis care platform formed with the aim to better serve medical cannabis patients’ needs and enhance the medical cannabis patients’ journey. MyMedi.ca is operated by Northern Green Canada Inc. and features a diverse portfolio of products and bilingual pharmacist-led patient support programs. MyMedi.ca also provides specialty services to distinct patient groups such as veterans and collaborates with public and private payers for adjudication and reimbursement. MyMedi.ca provides educational resources to the medical community to facilitate the incorporation of medical cannabis into health care regimens.
- Pharmaceutical pipeline: Leveraging Avicanna’s scientific platform, vertical integration, and real-world evidence, Avicanna has developed a pipeline of proprietary, indication-specific cannabinoid-based candidates that are in various stages of clinical development. These cannabinoid-based candidates aim to address unmet needs in the areas of dermatology, chronic pain, and various neurological disorders.
- Active pharmaceutical ingredients (Aureus Santa Marta™): Active pharmaceutical ingredients supplied by the Company’s majority owned subsidiary Santa Marta Golden Hemp SAS (“SMGH”) is a commercial-stage business dedicated to providing various forms of high-quality CBD, THC and CBG to the Company’s international partners for use in the development and production of food, cosmetics, medical, and pharmaceutical products. SMGH also forms part of the Company’s supply chain and is a source of reliable input products for its consumer retail, medical cannabis, and pharmaceutical products globally.
SOURCE Avicanna Inc.
Stay Connected
For more information about Avicanna, visit our website or contact Ivana Maric by email at ir@avicanna.com.
Cautionary Note Regarding Forward-Looking Information and Statements
This news release contains “forward-looking information” within the meaning of applicable securities laws. Forward-looking information contained in this news release may be identified by the use of words such as, “may”, “would”, “could”, “will”, “likely”, “expect”, “anticipate”, “believe”, “intend”, “plan”, “forecast”, “project”, “estimate”, “outlook” and other similar expressions. Forward-looking information contained in this news release includes, without limitation, statements related to the Company’s future business operations, the opinions or beliefs of management and future business goals. Although the Company believes that the expectations and assumptions on which such forward looking information is based are reasonable, undue reliance should not be placed on the forward-looking information because the Company can give no assurance that they will prove to be correct. Actual results and developments may differ materially from those contemplated by these statements. Forward-looking information is subject to a variety of risks and uncertainties that could cause actual events or results to differ materially from those projected in the forward-looking information. Such risks and uncertainties include, but are not limited to current and future market conditions, including the market price of the common shares of the Company, and the risk factors set out in the Company’s annual information form dated March 31, 2026, filed with the Canadian securities regulators and available under the Company’s profile on SEDAR+ at www.sedarplus.ca. The statements in this news release are made as of the date of this release. The Company disclaims any intent or obligation to update any forward-looking information, whether as a result of new information, future events or results or otherwise, other than as required by applicable securities laws.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/6f98d5a4-c37e-4f97-9684-cb12bc90d2fd



Avicanna Reports Full Year 2025 Audited Financial Statements
Achieved positive adjusted EBITDA in Q4 2025 and near break-even $(0.29) million for the full yearAdvancements across business units, R&D, and clinical development to support scale and international growth
© 2026 Canjex Publishing Ltd. All rights reserved.